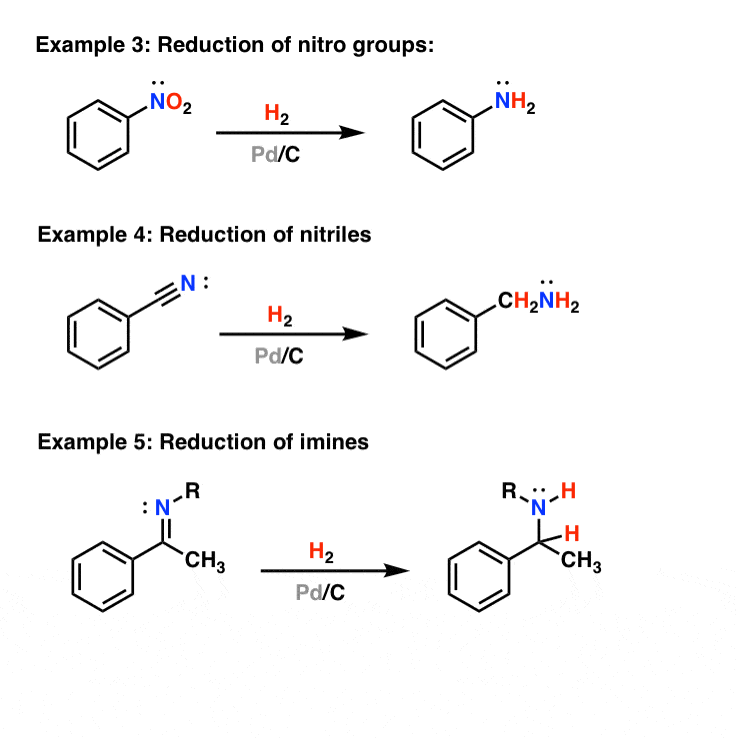
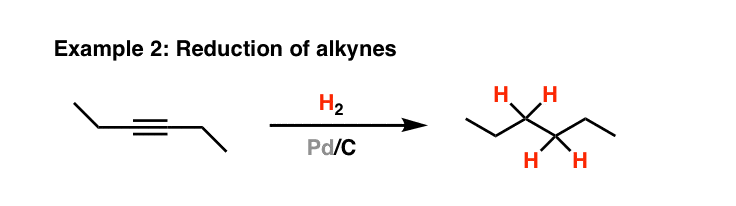
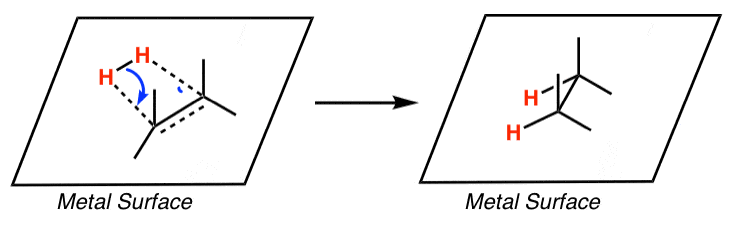
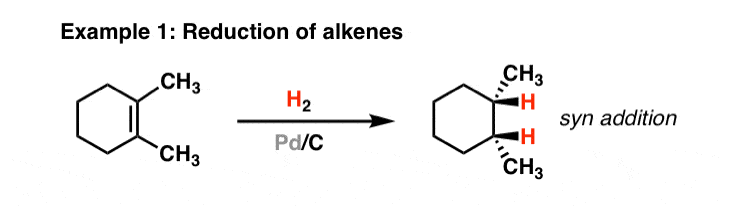
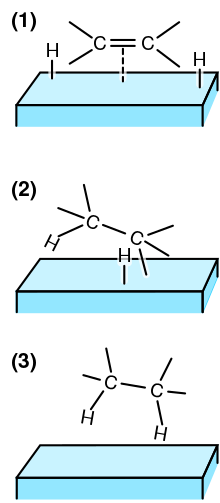
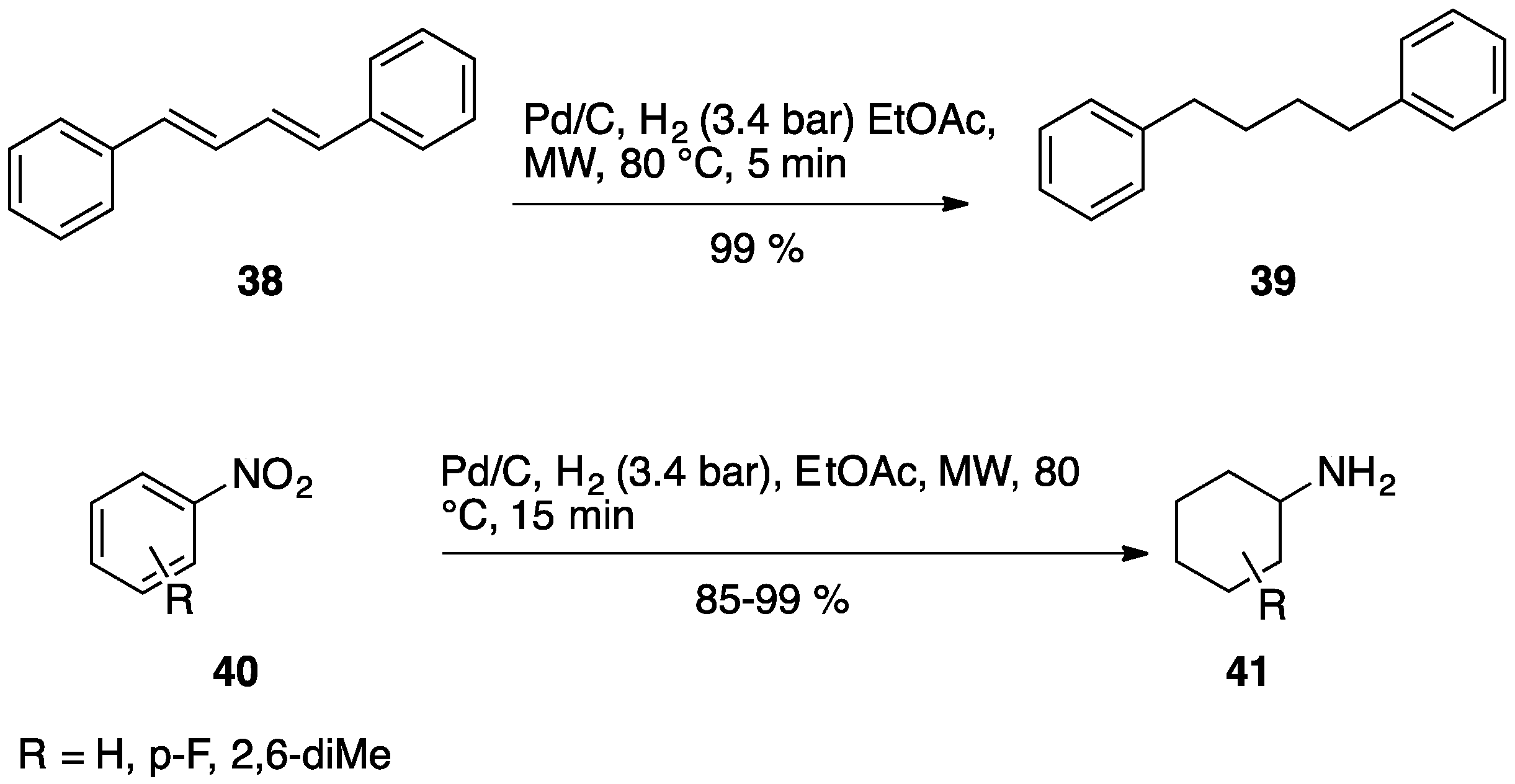
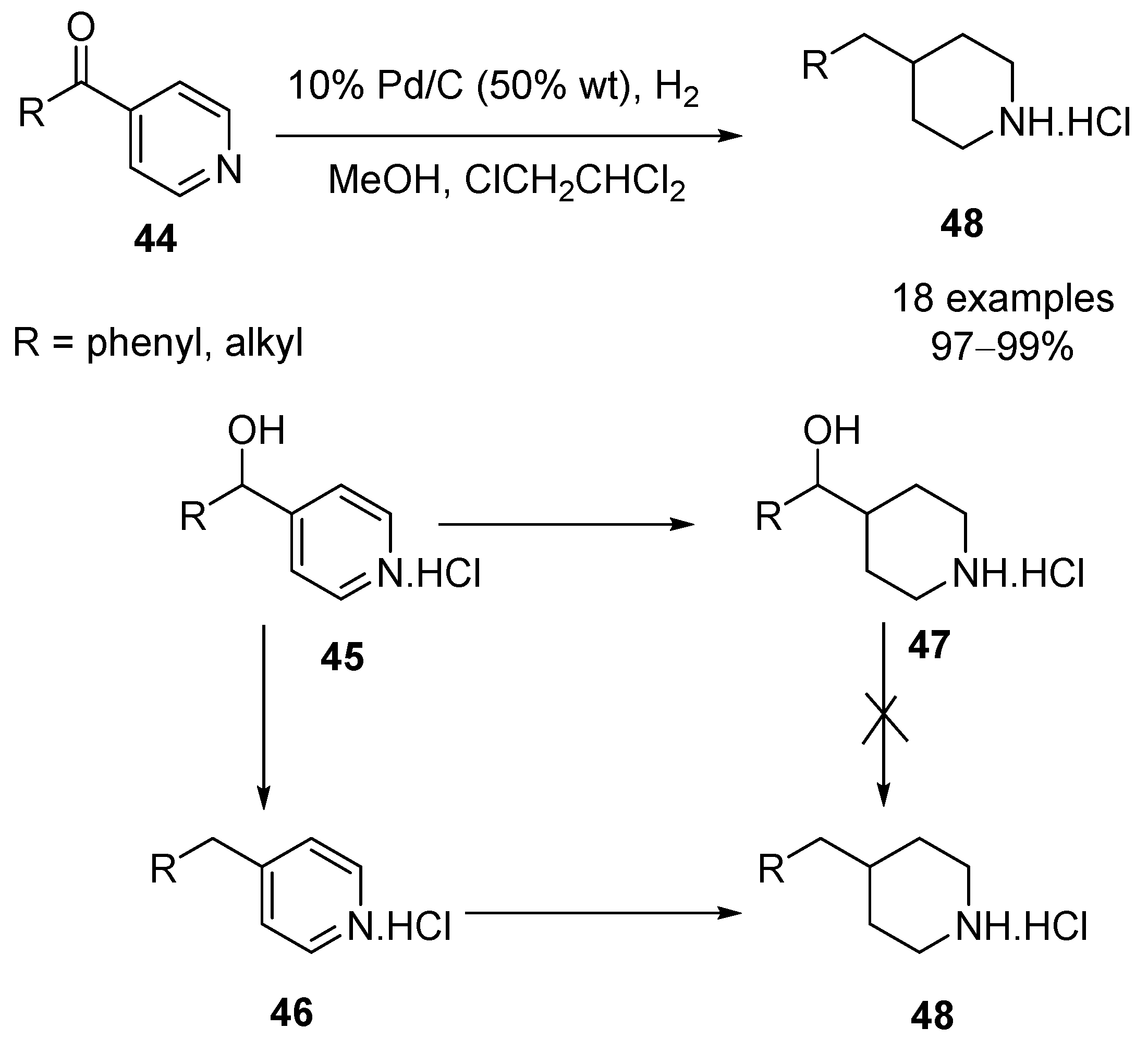
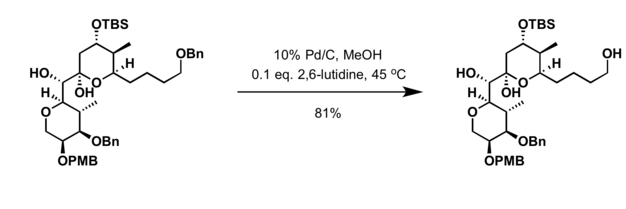
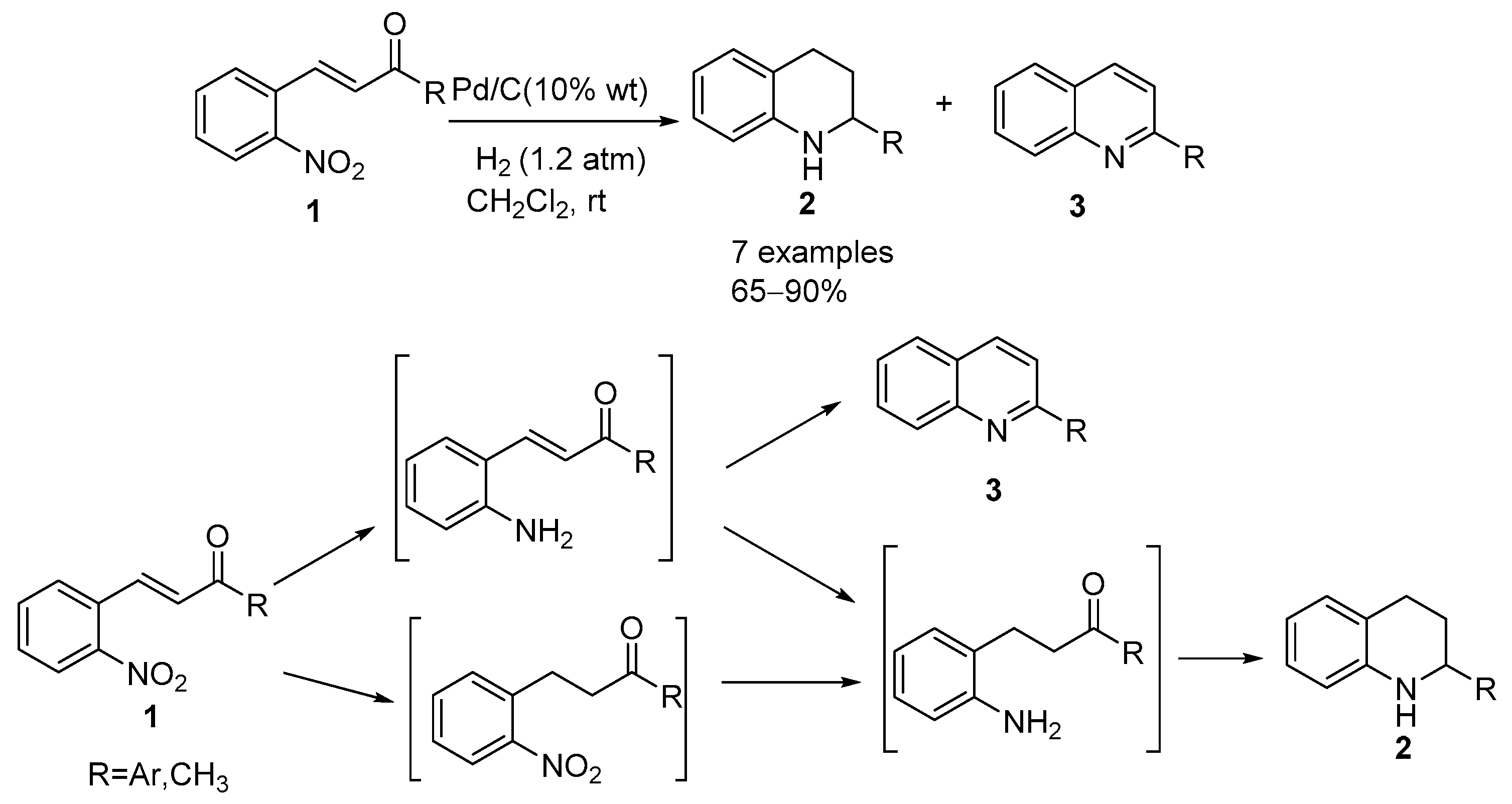
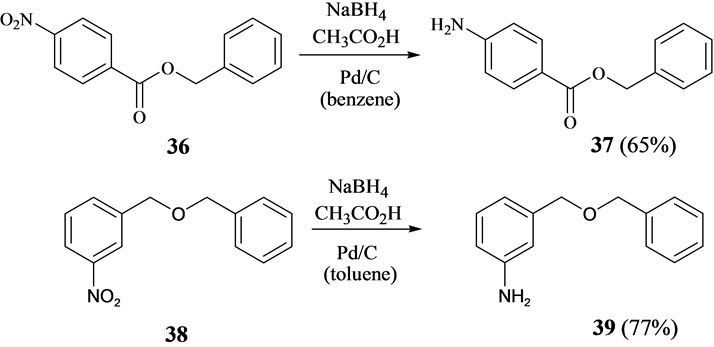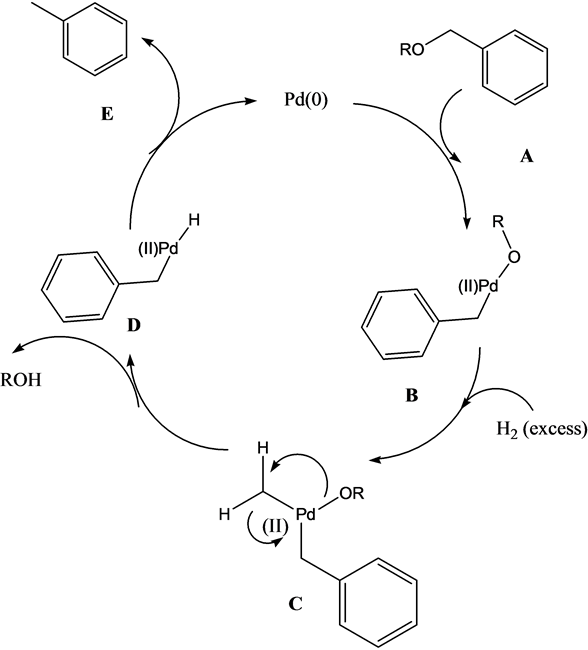Recyclability study of Pd/C Catalyst. Reaction conditions: 1a (1 mmol),... | Download Scientific Diagram
![Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/us/category/images/00074-img02.png)
Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation
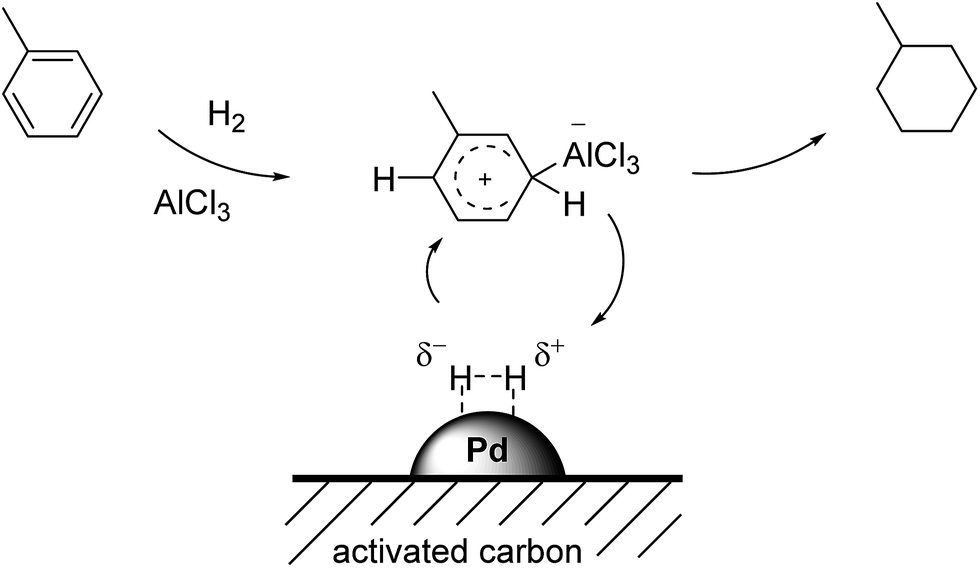
Coating of Pd/C catalysts with Lewis-acidic ionic liquids and liquid coordination complexes – SCILL induced activity enhancement in arene hydrogenatio ... - RSC Advances (RSC Publishing) DOI:10.1039/C7RA03295A

Catalytic hydrogenation of sulfur-containing nitrobenzene over Pd/C catalysts: In situ sulfidation of Pd/C for the preparation of PdxSy catalysts - ScienceDirect

Pd/C: An Efficient, Heterogeneous and Reusable Catalyst for Phosphane‐Free Carbonylative Suzuki Coupling Reactions of Aryl and Heteroaryl Iodides - Khedkar - 2010 - European Journal of Organic Chemistry - Wiley Online Library

Ammonium Formate/Palladium on Carbon: A Versatile System for Catalytic Hydrogen Transfer Reductions of Carbon-Carbon Double Bonds
![Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/us/category/images/00074-img07.png)
Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation
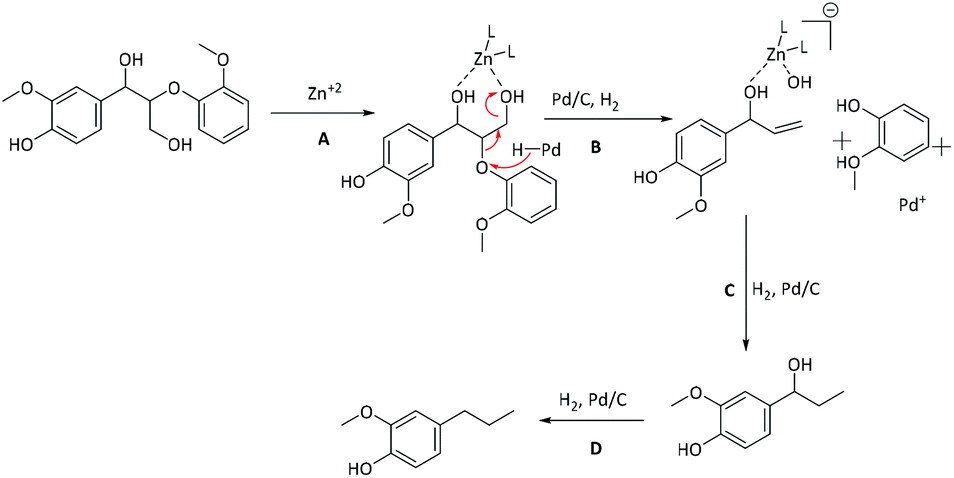
Mechanistic investigation of the Zn/Pd/C catalyzed cleavage and hydrodeoxygenation of lignin - Green Chemistry (RSC Publishing) DOI:10.1039/C5GC01325A
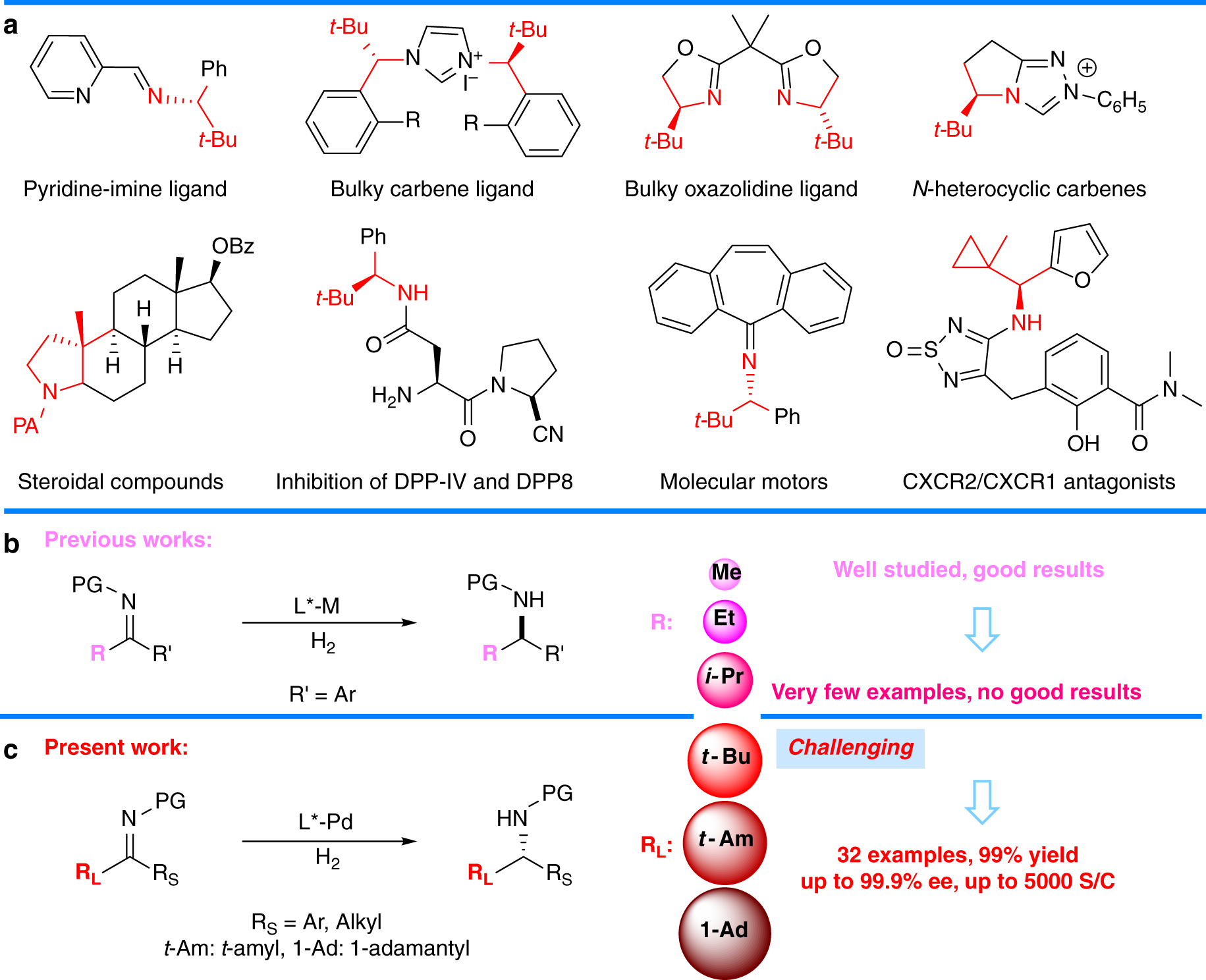
Pd(OAc)2-catalyzed asymmetric hydrogenation of sterically hindered N-tosylimines | Nature Communications