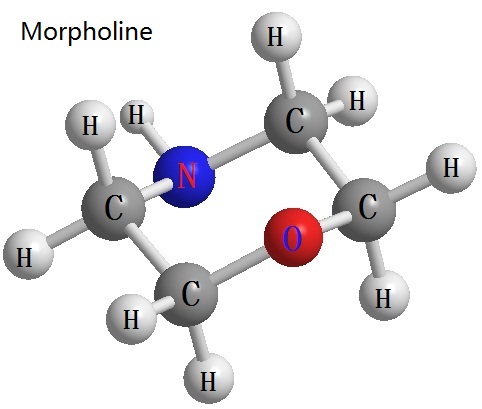
Figure 1 from SYNTHESIS AND BIOLOGICAL EVALUATION OF (3S)-3-(4- METHOXYMETHOXY) BENZYL) MORPHOLINE DERIVATIVES FROM L-TYROSINE | Semantic Scholar

Morpholine As a Scaffold in Medicinal Chemistry: An Update on Synthetic Strategies - Tzara - 2020 - ChemMedChem - Wiley Online Library
LC-MS/MS reveals the formation of aldehydes and iminium reactive intermediates in foretinib metabolism: phase I metabolic profil

Morpholine as a privileged structure: A review on the medicinal chemistry and pharmacological activity of morpholine containing bioactive molecules - Kourounakis - 2020 - Medicinal Research Reviews - Wiley Online Library

Figure 1 from SYNTHESIS AND BIOLOGICAL EVALUATION OF (3S)-3-(4- METHOXYMETHOXY) BENZYL) MORPHOLINE DERIVATIVES FROM L-TYROSINE | Semantic Scholar

E11 pancreas cultured for 6 days in the presence of morpholine-ring antisense against GIP, GIPR, or control. E11 pancreas cultured for 6 days in the presence. - ppt download
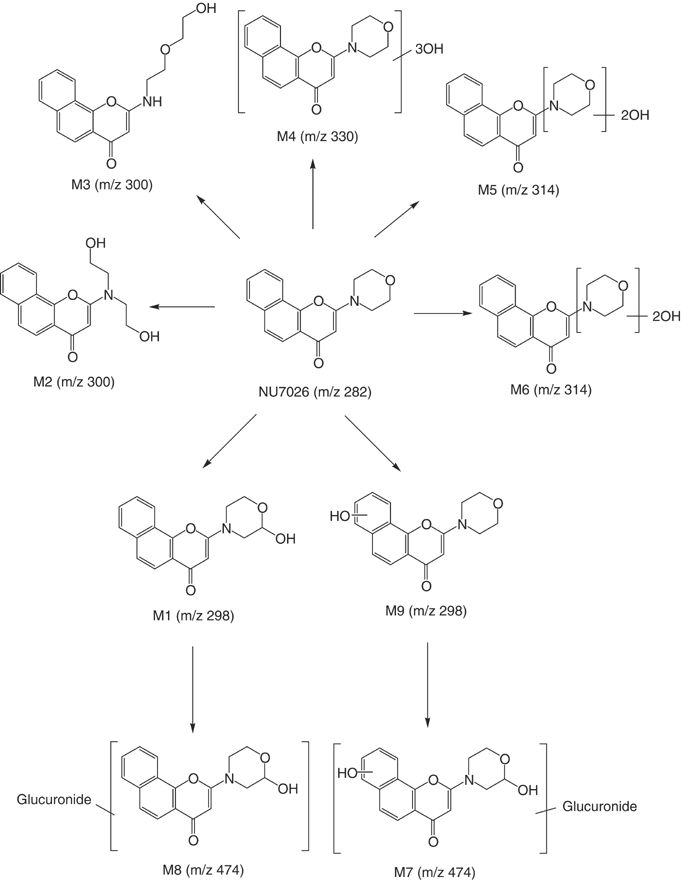
Preclinical pharmacokinetics and metabolism of a novel prototype DNA-PK inhibitor NU7026 | British Journal of Cancer

Synthesis and antiviral study of 4-(7,7-dimethyl-4-(piperazin-1-yl)-5,6,7,8-tetrahydroquinazolin-2-yl) morpholine derivatives | SpringerLink
Rocuronium Has a Suppressive Effect on Platelet Function via the P2Y12 Receptor Pathway In Vitro That Is Not Reversed by Sugamma

Common degradative pathways of morpholine, thiomorpholine, and piperidine by Mycobacterium aurum MO1: evidence from (1)H-nuclear magnetic resonance and ionspray mass spectrometry performed directly on the incubation medium. - Abstract - Europe PMC

Morpholine as a privileged structure: A review on the medicinal chemistry and pharmacological activity of morpholine containing bioactive molecules - Kourounakis - 2020 - Medicinal Research Reviews - Wiley Online Library
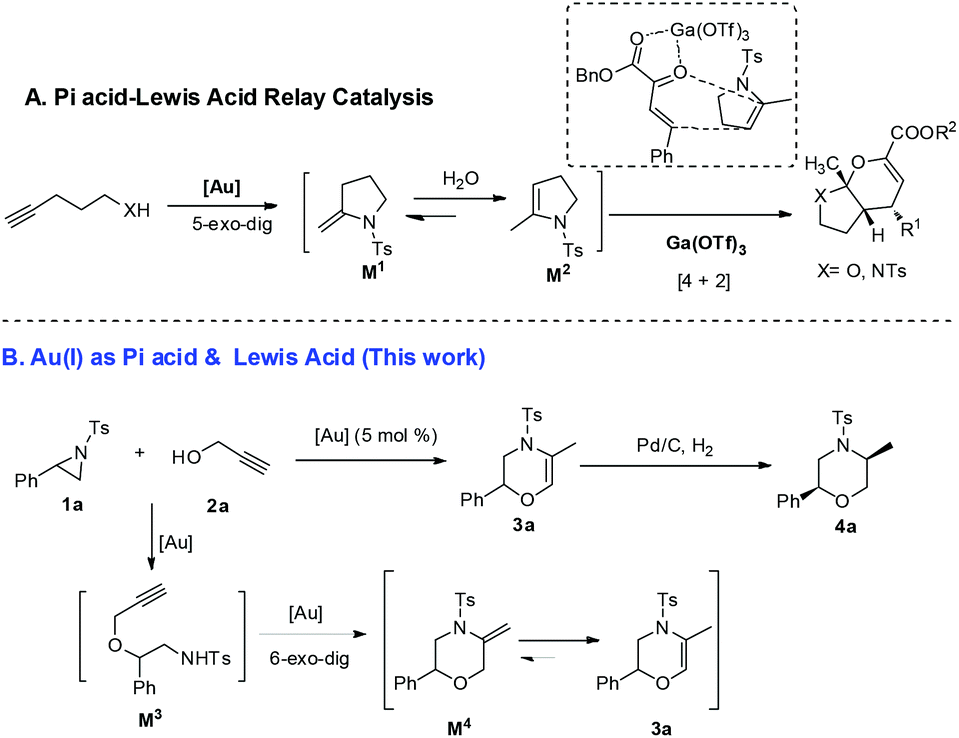
Breaking aziridines to construct morpholines with a gold(i)-catalyzed tandem ring-opening and cycloisomerization reaction - Organic & Biomolecular Chemistry (RSC Publishing)
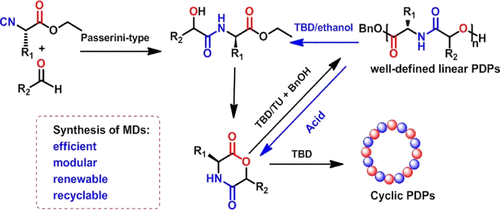
Synthesis and Controlled Organobase-Catalyzed Ring-Opening Polymerization of Morpholine-2,5-Dione Derivatives and Monomer Recovery by Acid-Catalyzed Degradation of the Polymers,Macromolecules - X-MOL