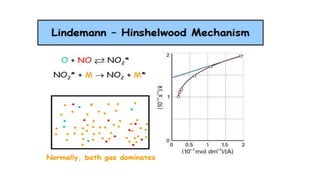
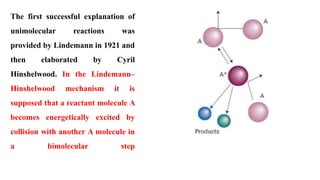
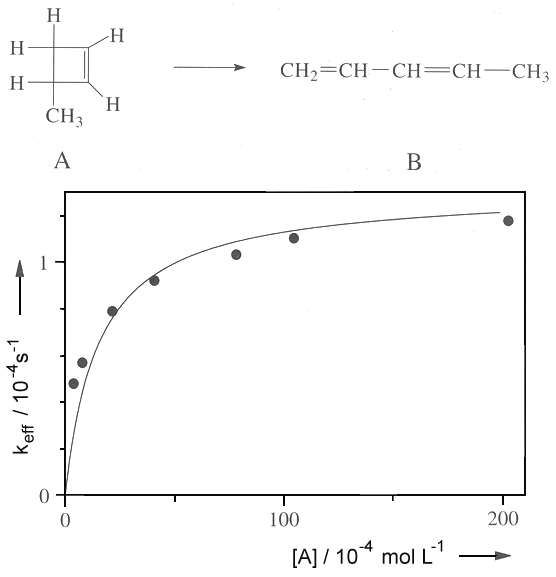
SOLVED:Outline how the steady state approximation can be applied to the Lindemann-Hinshelwood mechanism for a unimolecular decomposition reaction. b) Using the steady state approximation method, and showing all working, use the following

How/Why have the arranged the Lindemann-Hinshelwood mechanism like this for 2 pressures (3rd line? : r/chemhelp

Chemistry 232 Complex Reaction Mechanisms. Lindemann-Hinshelwood Mechanism An early attempt to explain the kinetics of complex reactions. Mechanism Rate. - ppt download

SOLVED:4) Partial pressures are frequently used instead of molar concentrations for reactions of gases that follows the ideal gas law. Consider the modified Lindemann-Hinshelwood mechanism that is employed for unimolecular reactions in