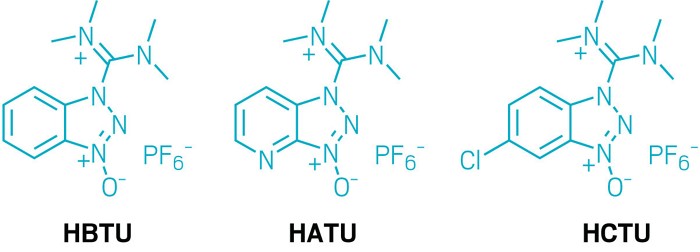
Propylphosphonic Anhydride (T3P®) as Coupling Reagent for Solid‐Phase Peptide Synthesis - Al Musaimi - 2021 - ChemistrySelect - Wiley Online Library

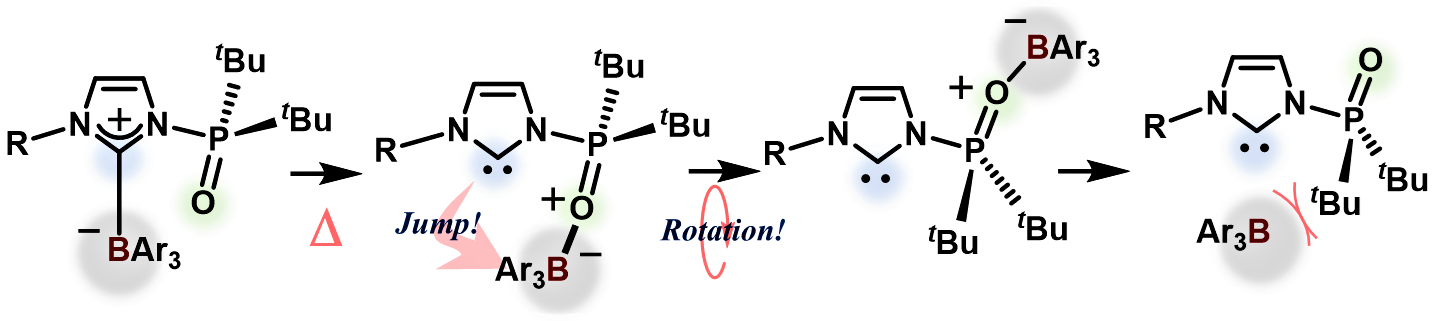
Rearrangement of the Active Ester Intermediate During HOBt/EDC Amide Coupling - Mahmoud - 2005 - European Journal of Inorganic Chemistry - Wiley Online Library
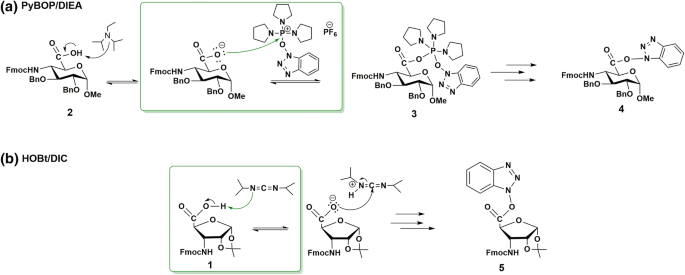
α/β-Chimera peptide synthesis with cyclic β-sugar amino acids: the efficient coupling protocol | SpringerLink

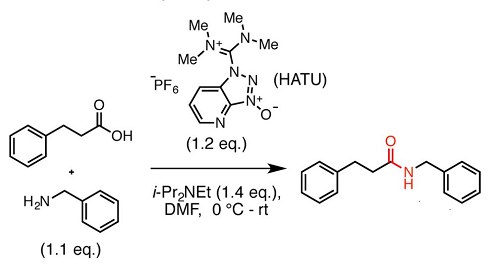
Synthesis of amide derivatives for electron deficient amines and functionalized carboxylic acids using EDC and DMAP and a catalytic amount of HOBt as the coupling reagents - ScienceDirect

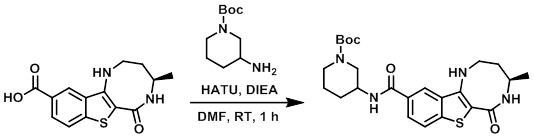
Thirteen decades of peptide synthesis: key developments in solid phase peptide synthesis and amide bond formation utilized in peptide ligation | SpringerLink
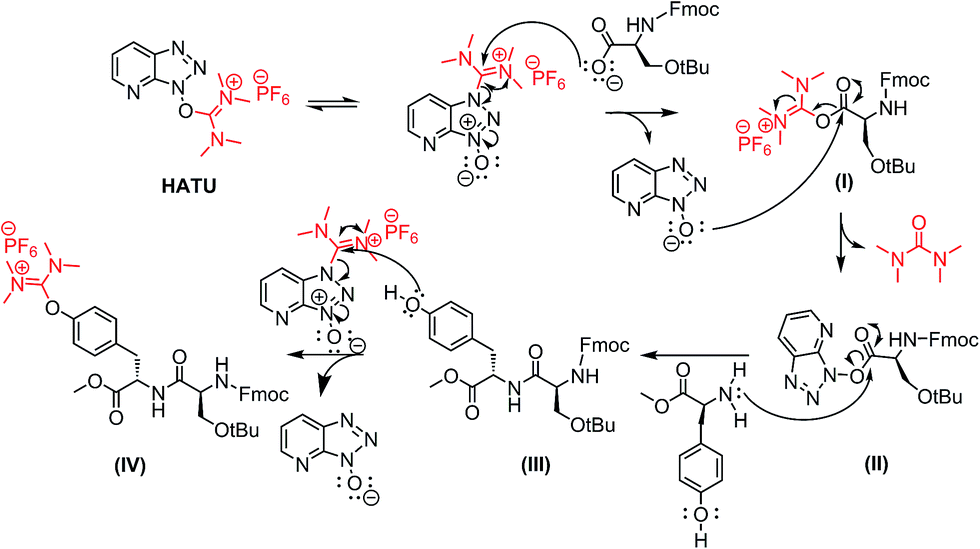
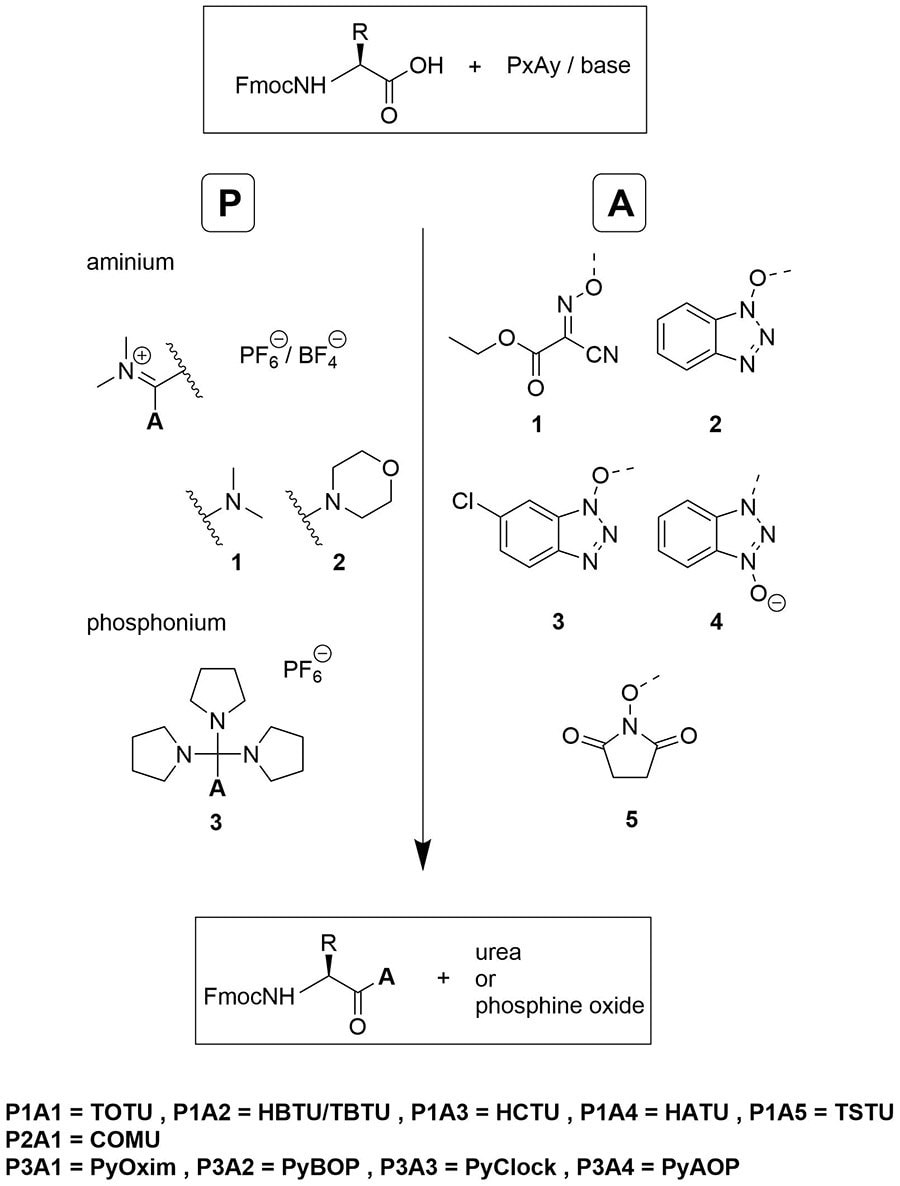
Unveiling and tackling guanidinium peptide coupling reagent side reactions towards the development of peptide-drug conjugates - RSC Advances (RSC Publishing) DOI:10.1039/C7RA06655D
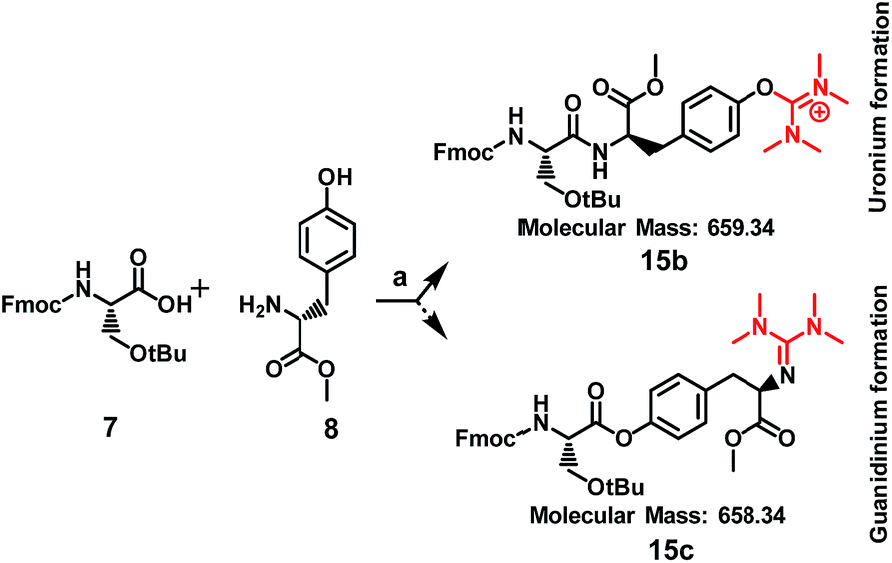
Unveiling and tackling guanidinium peptide coupling reagent side reactions towards the development of peptide-drug conjugates - RSC Advances (RSC Publishing) DOI:10.1039/C7RA06655D

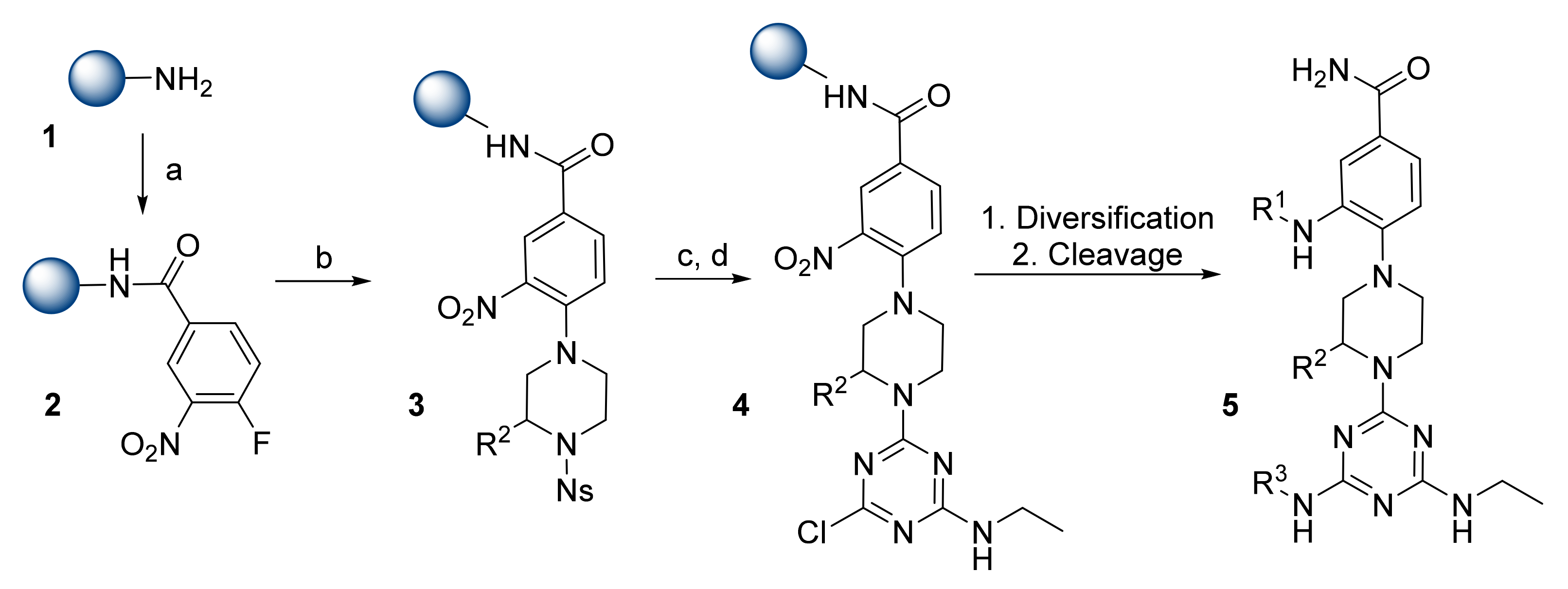
General synthesis of compounds 6-19. Reagents and conditions: a) HATU,... | Download Scientific Diagram

On resin click-chemistry-mediated synthesis of novel enkephalin analogues with potent anti-nociceptive activity | Scientific Reports