![Dimroth rearrangement-based synthesis of novel derivatives of [1,3]selenazolo[5,4-e][1,2,4]triazolo[1,5-c]pyrimidine as a new class of selenium-containing heterocyclic architecture | SpringerLink Dimroth rearrangement-based synthesis of novel derivatives of [1,3]selenazolo[5,4-e][1,2,4]triazolo[1,5-c]pyrimidine as a new class of selenium-containing heterocyclic architecture | SpringerLink](https://media.springernature.com/lw685/springer-static/image/art%3A10.1007%2Fs11030-021-10203-9/MediaObjects/11030_2021_10203_Fig9_HTML.png)
Dimroth rearrangement-based synthesis of novel derivatives of [1,3]selenazolo[5,4-e][1,2,4]triazolo[1,5-c]pyrimidine as a new class of selenium-containing heterocyclic architecture | SpringerLink

Recent Advances in the Dimroth Rearrangement: A Valuable Tool for the Synthesis of Heterocycles - ScienceDirect
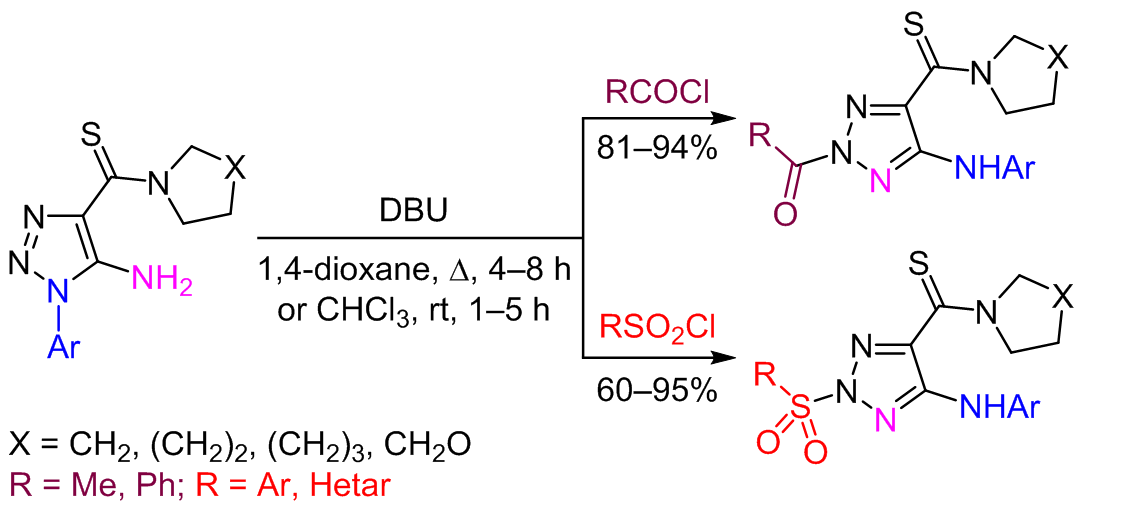
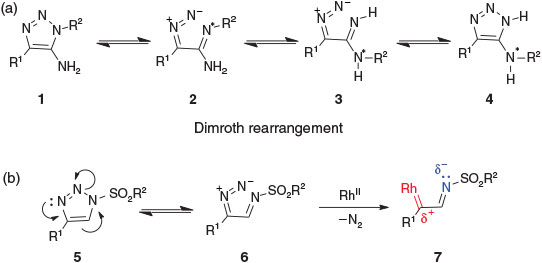
THE TANDEM DIMROTH REARRANGEMENT AND SULFONYLATION/ACYLATION AS REGIOSELECTIVE METHOD FOR THE SYNTHESIS OF 5-ARYLAMINO-2-SULFONYLAND 2-ACYL-5-ARYLAMINO-1,2,3-TRIAZOLE-4-CARBOTHIOAMIDES | Дианова | Chemistry of Heterocyclic Compounds

Recent Advances in the Dimroth Rearrangement: A Valuable Tool for the Synthesis of Heterocycles - ScienceDirect

Applications of Purine Ring Opening in the Synthesis of Imidazole, Pyrimidine, and New Purine Derivatives - Leškovskis - 2021 - European Journal of Organic Chemistry - Wiley Online Library

The Dimroth Rearrangement in the Synthesis of Condensed Pyrimidines – Structural Analogs of Antiviral Compounds - Abstract - Europe PMC

The Dimroth Rearrangement in the Synthesis of Condensed Pyrimidines – Structural Analogs of Antiviral Compounds | SpringerLink

Verdazyl Radicals as Substrates for Organic Synthesis: Unique Access to Tetrahydropyrazolotriazinones, Pyrazolotriazinones and Dihydrotetrazinylacrylonitriles - Chen - 2010 - European Journal of Organic Chemistry - Wiley Online Library
![Tunable regioselective synthesis of pyrazolo[3,4- d ]pyrimidine derivatives via aza-Wittig cyclization and dimroth-type rearrangement - RSC Advances (RSC Publishing) DOI:10.1039/C4RA15777J Tunable regioselective synthesis of pyrazolo[3,4- d ]pyrimidine derivatives via aza-Wittig cyclization and dimroth-type rearrangement - RSC Advances (RSC Publishing) DOI:10.1039/C4RA15777J](https://pubs.rsc.org/image/article/2015/RA/c4ra15777j/c4ra15777j-s3_hi-res.gif)
Tunable regioselective synthesis of pyrazolo[3,4- d ]pyrimidine derivatives via aza-Wittig cyclization and dimroth-type rearrangement - RSC Advances (RSC Publishing) DOI:10.1039/C4RA15777J
![Synthesis of new 3H-chromeno[2,3-d]pyrimidine-4,6(5H,7H)-diones via the tandem intramolecular Pinner/Dimroth rearrangement Synthesis of new 3H-chromeno[2,3-d]pyrimidine-4,6(5H,7H)-diones via the tandem intramolecular Pinner/Dimroth rearrangement](https://www.degruyter.com/document/doi/10.1515/hc-2017-0228/asset/graphic/j_hc-2017-0228_scheme_002.jpg)
Synthesis of new 3H-chromeno[2,3-d]pyrimidine-4,6(5H,7H)-diones via the tandem intramolecular Pinner/Dimroth rearrangement

An alternative synthesis of Vandetanib (Caprelsa™) via a microwave accelerated Dimroth rearrangement. - Abstract - Europe PMC

Recent Advances in the Dimroth Rearrangement: A Valuable Tool for the Synthesis of Heterocycles - ScienceDirect