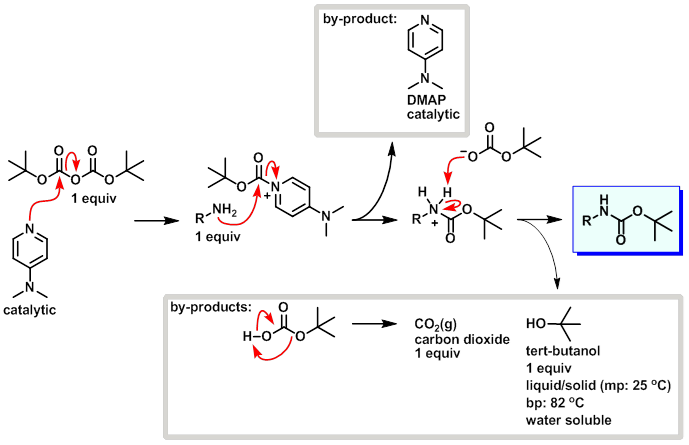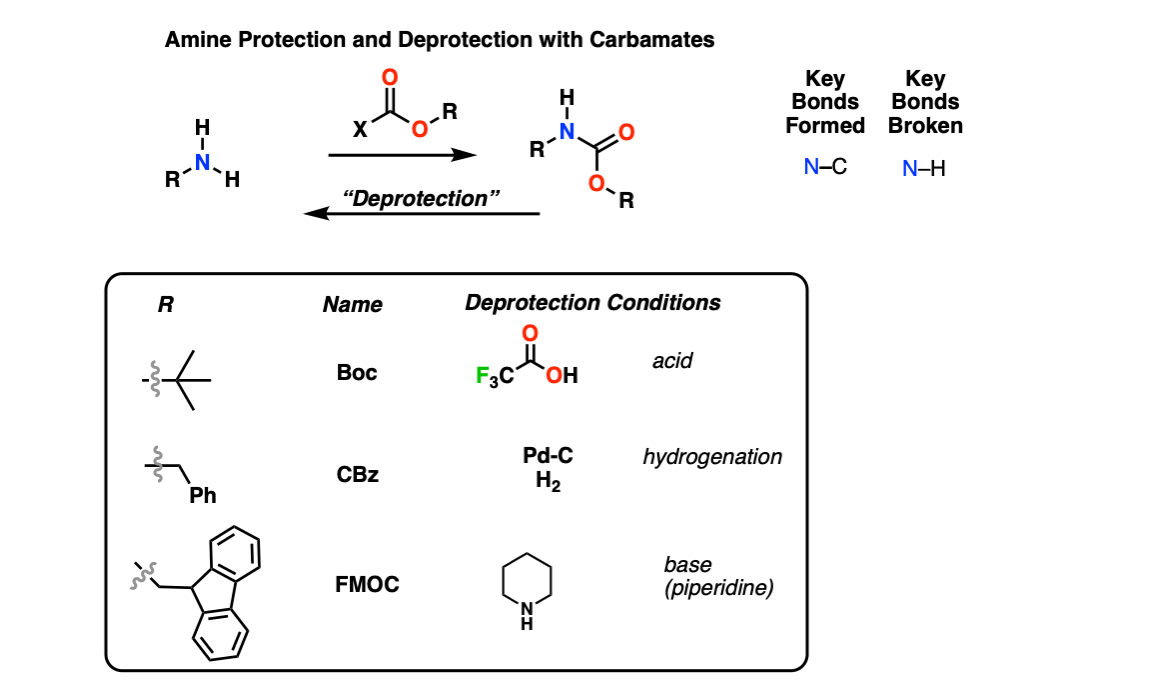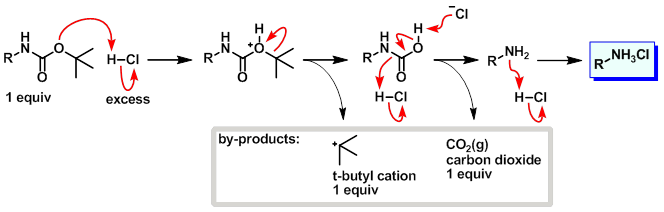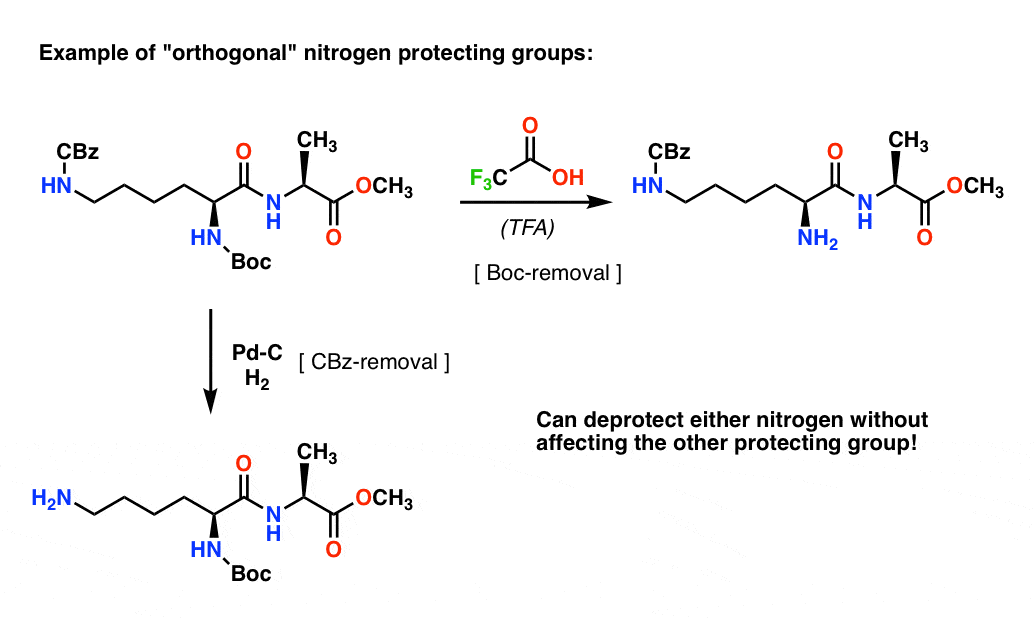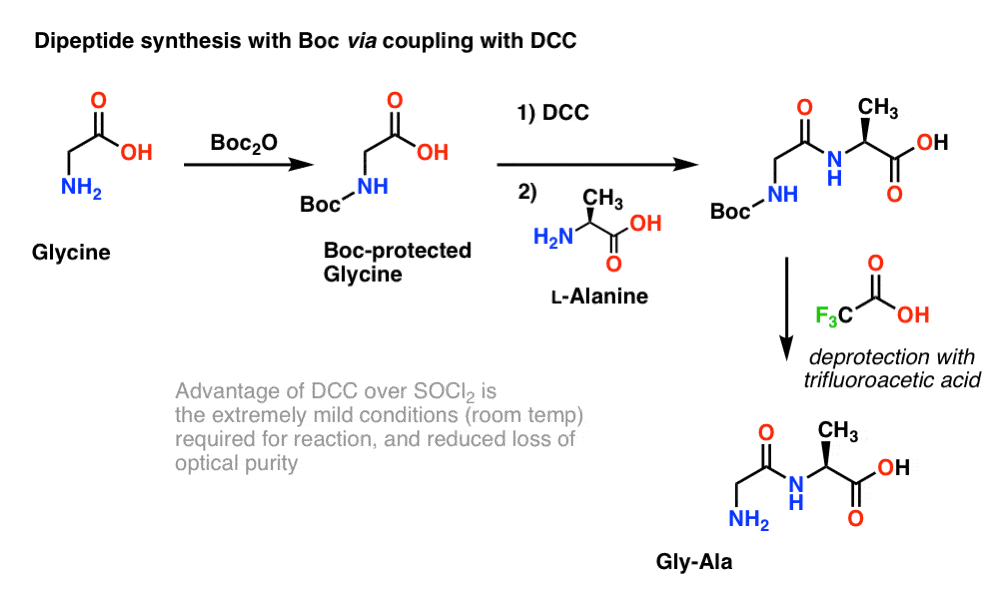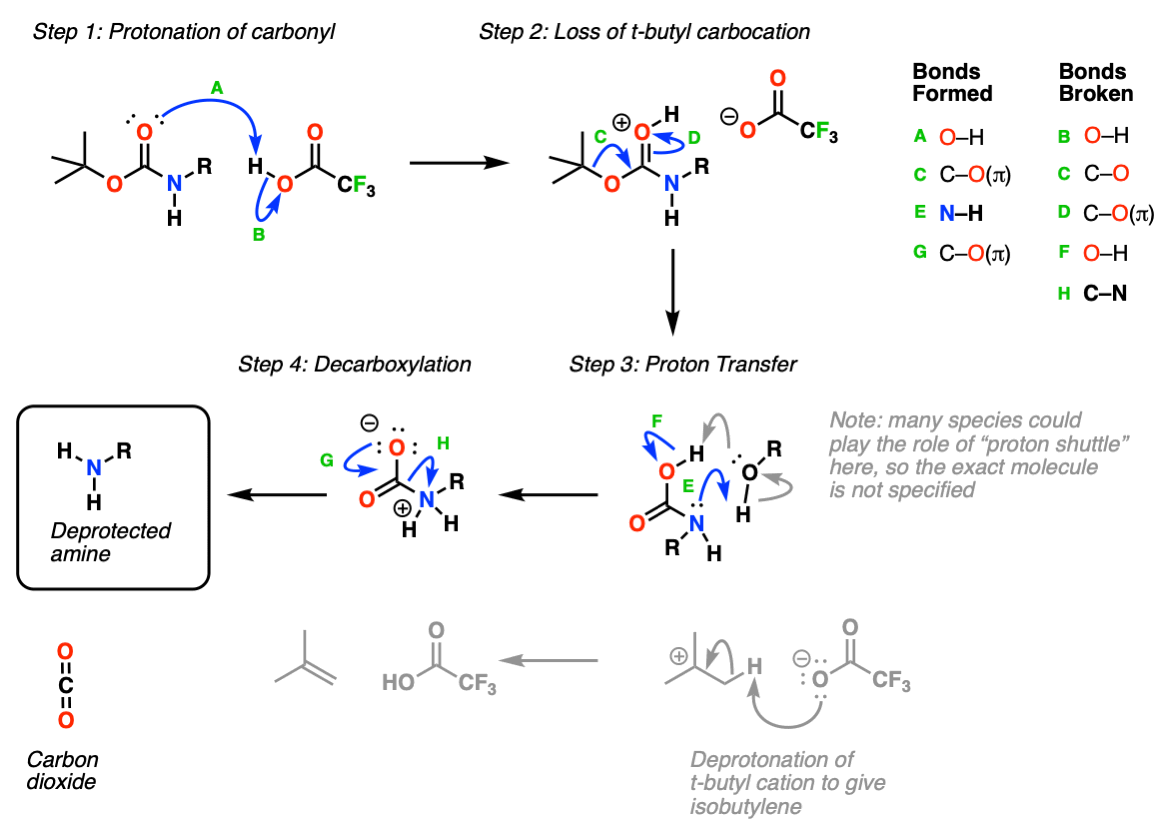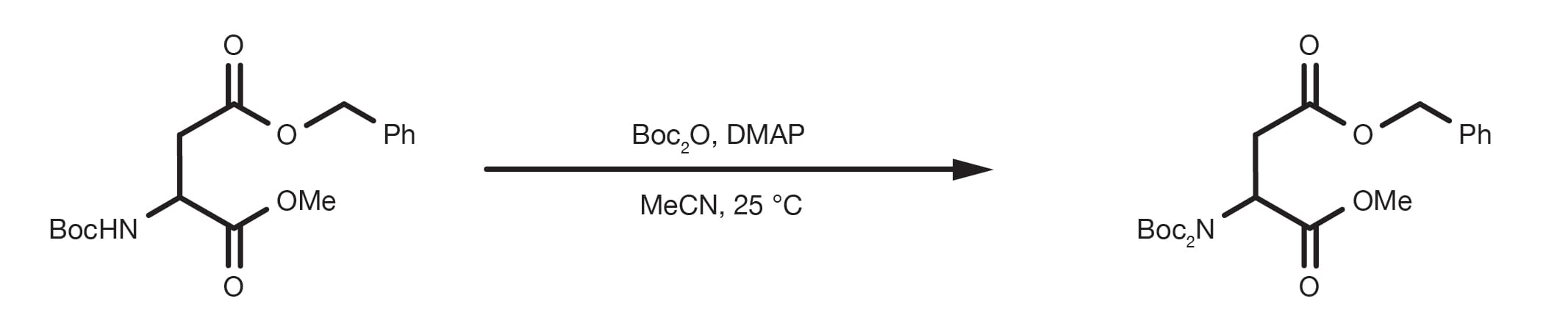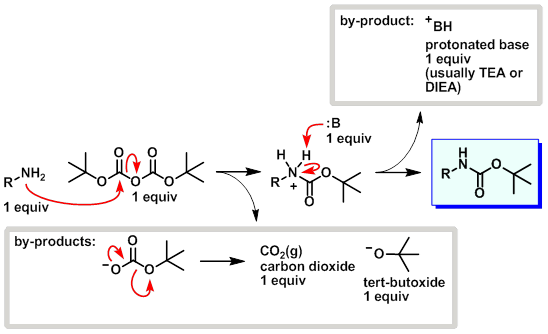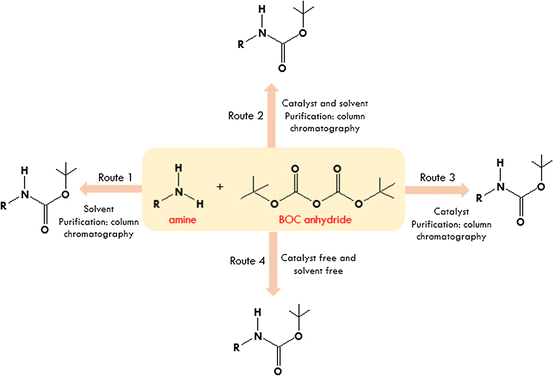
Efficient and expeditious chemoselective BOC protection of amines in catalyst and solvent-free media | SpringerLink

Efficient and expeditious chemoselective BOC protection of amines in catalyst and solvent-free media | SpringerLink

A one-pot selective synthesis of N-Boc protected secondary amines: tandem direct reductive amination/N-Boc protection - ScienceDirect
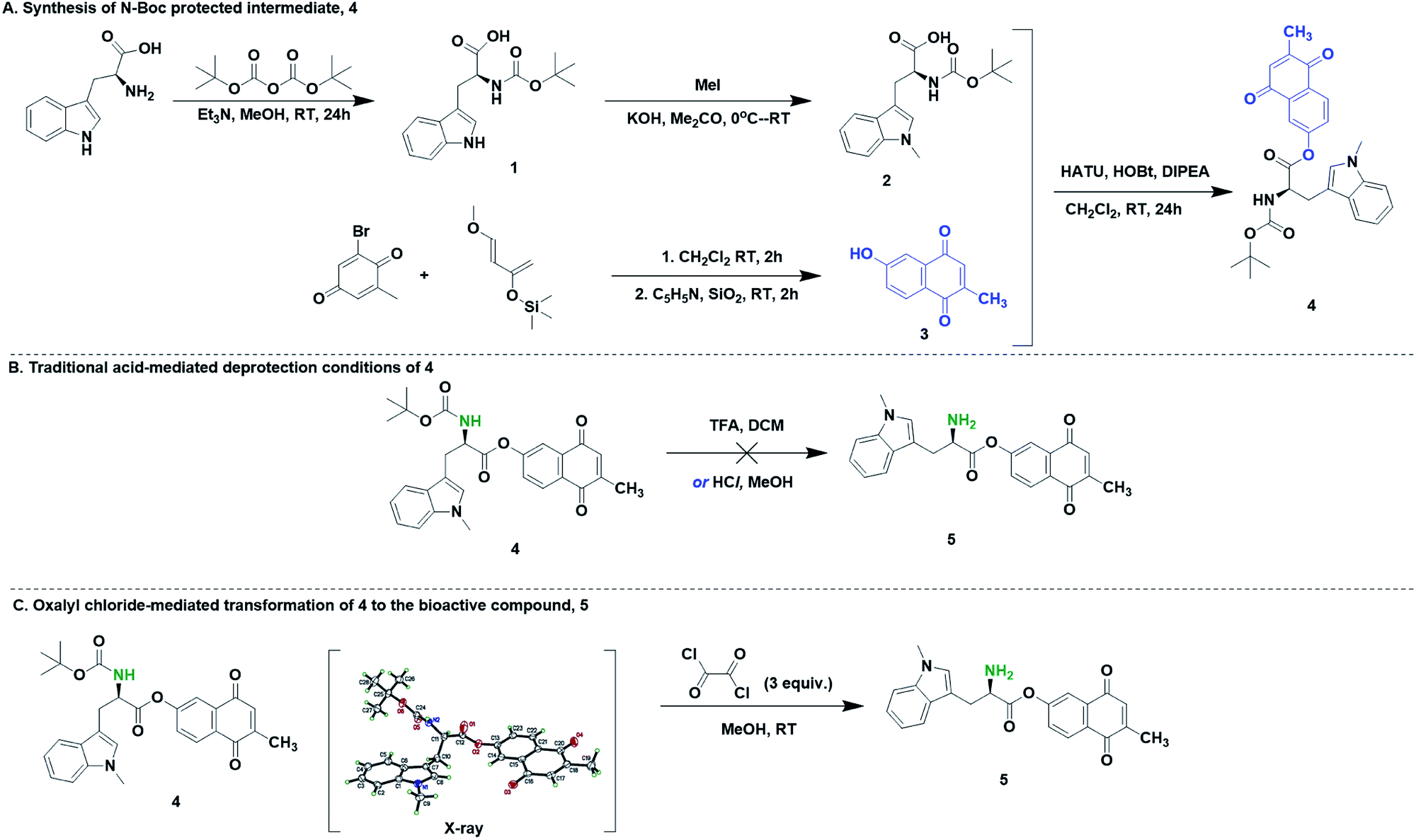
Mild deprotection of the N-tert -butyloxycarbonyl ( N -Boc) group using oxalyl chloride - RSC Advances (RSC Publishing) DOI:10.1039/D0RA04110F

Deprotection of N‐tert‐Butoxycarbonyl (Boc) Protected Functionalized Heteroarenes via Addition–Elimination with 3‐Methoxypropylamine - Gulledge - 2020 - European Journal of Organic Chemistry - Wiley Online Library